Diseases & Treatments.
We are committed to you the patient, and your family. Every step of our process in your care is dedicated to the point of giving you the best modern medicine can provide in a manner that is in keeping with your life, health and circumstances.
Peripheral Vascular Disease (PVD)
Blockages in leg arteries can cause severe pain with walking and wounds.
Spider & Varicose Veins
Stretching can weaken the walls of the veins and damage their valves causing blood back-up.
Deep Vein Thrombosis (DVT)
Venous thrombosis commonly presents as DVT of the legs and pulmonary embolism.
Mesenteric Ischemia
A fold of membranous tissue that attaches to the intestinal tract.
Abdominal Aortic Aneurysm (AAA)
Treatment includes the deployment of stent grafts with minimal recovery time.
Renal Artery Stenosis (RAS)
Managed through a combination of lifestyle changes, medication, and surgical intervention.
Wound Care
Treatment goals include increasing recovery speed, and fully healing the wound.
Hyperbaric Oxygen Therapy (HBOT)
An adjunctive therapy for acute traumatic and ischemic injuries, such as crush injuries.
Carotid Disease
Blockages resulting in decreased blood flow and oxygen supply to the brain.
Aortic Aneurysm Ultrasound
For men aged 65-75 with smoking history, or with close relative who required aortic surgery.
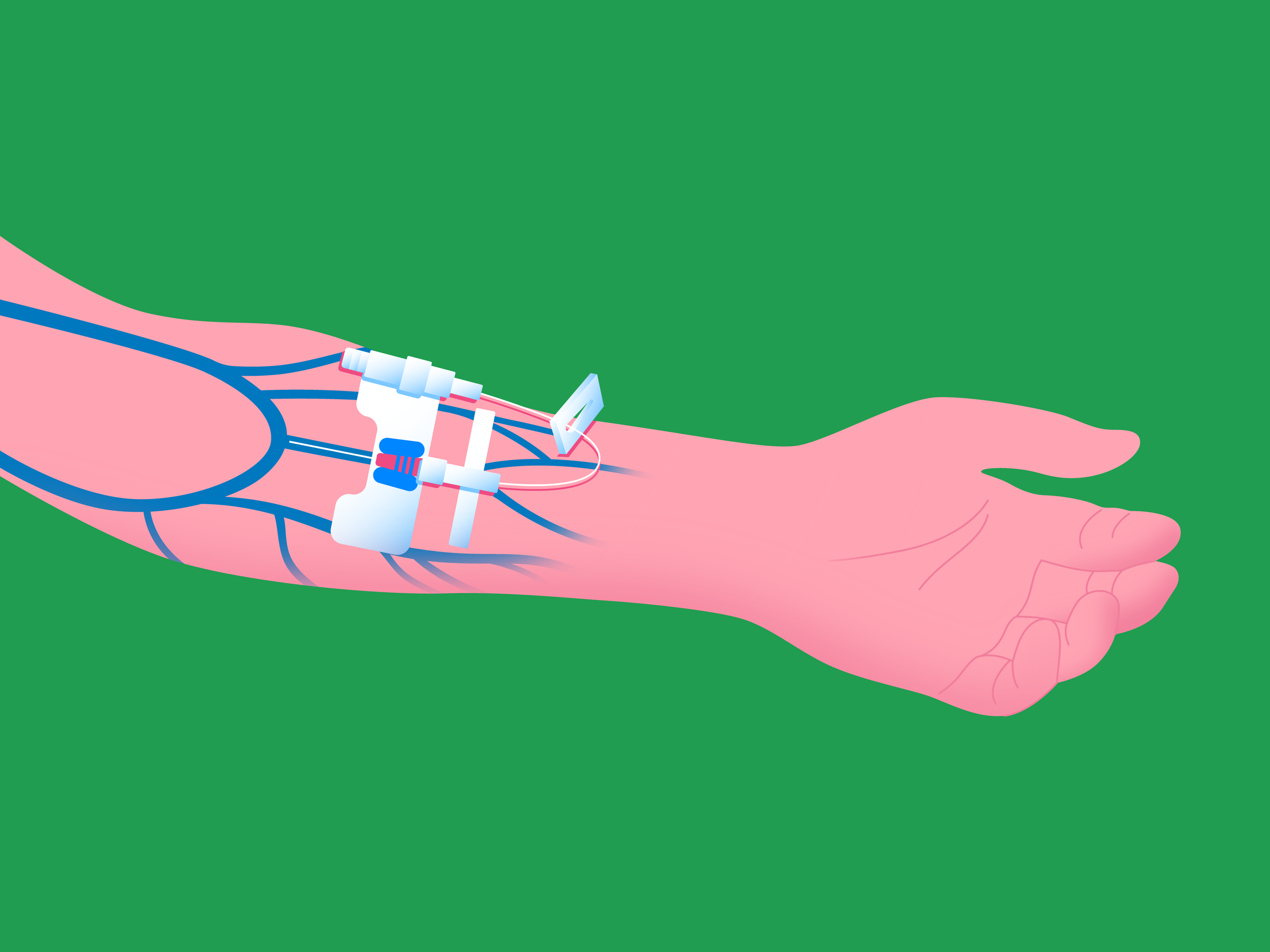
Dialysis Access
Dialysis requires temporary or long-term blood access sites via minimally invasive surgery.
Chronic Pelvic Pain Syndrom (CPPS)
Pain below your belly button and between your hips that lasts 6 months or longer.
Raynaud’s Syndrome
Managed via medication, minimally invasive surgery, or in combination.
Thoracic Aortic Aneurysm (TAA)
Stent grafts are deployed through bilateral groin regions with minimal recovery time.
Lipid Disorders
Among the most common risk factors for heart disease and stroke.
May-Thurner Syndrome (MTS)
Treated with a combination of medication and minimally invasive surgery.
Smoking Cessation
Accomplished through strength of will, nicotine replacement, medication, or in combination.
Pulmonary Embolism (PE)
Managed via compression stockings therapy, minimally invasive surgery, or in combination.
Thoracic Outlet Syndrome (TOS)
Compression of blood vessels or nerves in the space between your collarbone and first rib.
Antibiotics
Medications that either kill bacteria directly or stop them from multiplying.
Venous Venography
Imaging process for evaluating the structure and functionality of veins.
Vascular Screening
This treatment can be helpful for identifying problems early on, before they become serious.
Antiplatelet Therapy
Platelets are cells in the blood that clump together to begin the clotting process.
Diabetic Retinopathy (DR)
Main cause of impaired vision for those between the ages of 25-74.
Atrial Fibrillation (AF)
A quivering atrium that lacks ability to drive blood into the ventricles for effective output.
Carotid Ultrasound
Duplex ultrasound for structure and Doppler assessment of blood flow.
RAPID® Technology
Vessel access from the foot, allowing for quicker recovery, shorter procedure times, and better results.
In-home appointment
Vascular Health Clinics provides Michigan state vascular evaluation and management in the comforts of your own home. Our specialists provide in-home consultation and evaluation for patients who have difficulty with travel.